 Tyrosine, threonine, even lysine have hydrophobic parts while being capable of making hydrogen bonds. This is not because proline is polar but because it is a helix and strand "breaker", so it is often found in turns, which are mostly on the surface of the protein.Īlso, you should know that there is no all-or-nothing classification of every amino acid side chain.  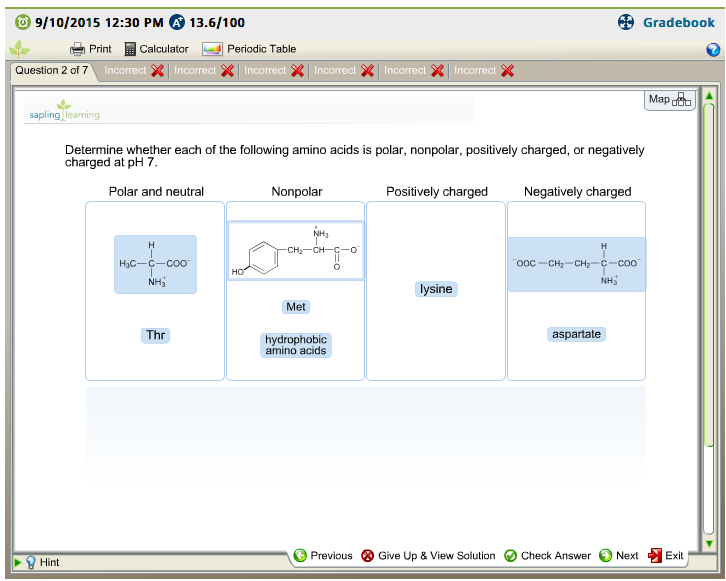
If you study where proline is mostly located with a folded protein, you find it is on the surface. Similarly confusing, proline is sometimes classified as polar amino acid however, its side chain is made exclusively of carbon and hydrogen atoms. The classification of amino acids is sometimes used as proxy of the tendency of amino acid side chains to be located on the surface or in the interior of a protein. This explains why methionine, the other sulfur-containing amino acid, is classified as hydrophobic. Protonated cysteine is incapable of making conventional hydrogen bonds, and the electronegativity of carbon and sulfur are quite similar. Cysteines that form disulfide bonds often are at or near the surface of the protein once the disulfide is formed, you have a quite hydrophobic group. In cysteine proteases, it acts as a nucleophile (and its surrounding often makes it more prone to deprotonation compared to a netural aqueous environment). 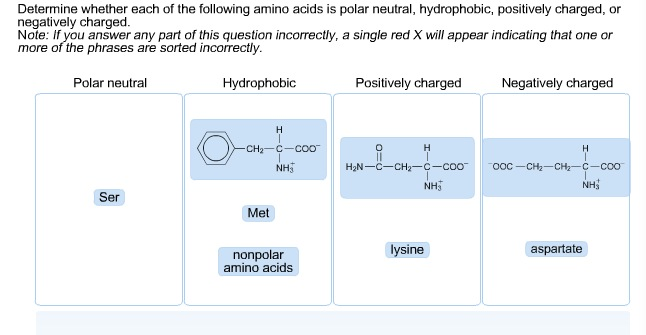
What you should know is that cysteine has different roles in proteins. If you search for amino acid classification, you will find that there is no agreement on how to classify cysteine. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
